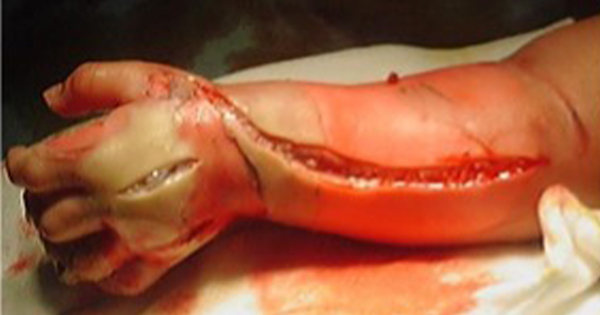
This document contains a series of case reports describing the use of ACTISORB® (Systagenix) in patients with a range of wound types. All patients were treated for a minimum of 2–4 weeks and the decision to continue with ACTISORB® was based on continual assessment. A formal assessment was performed weekly, although in some cases dressing changes were carried out more frequently.
Supported by Systagenix – An Acelity Company